Nats 101, Section 41, 42H- Introduction to Global
Change
Description and Exercises on:
This self-study begins with background information about converting between
units, cancelling of units in equations, conversion to and from scientific
notation, and interpreting graphs. If you feel comfortable with these areas,
then you may skip to the exercises section.
Converting
from one system of measurement to another is like translating from one language
to another, except that we use equations to make the change. The examples below
demonstrate the procedure.
Example
1: You have borrowed your friend's old 1971 Toyota Landcruiser for a scuba
trip to
X Km/hr * (1
mi/1.609 Km) = Y mi/hr.
(Read this as "the number
of kilometers per hour, times 1 mile per 1.609 kilometers, equals the number of
miles per hour.")
Solution:
120km/hr x 1mi/1.609km = 74.58 miles/hr, so you can drive at just under 75
miles per hour and not get pulled over (assuming the speedometer is accurate!)
Example
2: You are at a store buying a microwave oven for your apartment. It must
fit on the kitchen counter under a cabinet in a space you measured to be 21in
wide by 15in high by 19in deep. The measurements on the microwave you want, are
in metric units. Oh No! Will a microwave oven 45cm wide by 39cm high by 43cm
deep fit on your counter?
We know
from the conversions table that 1 inch = 2.54 centimeters, and 1cm = 0.3937
inches, so we can solve this problem going either direction. Here are the ways
to set up the equations:
(1)
Solution for space: inches to centimeters (need space larger than microwave
size)
X
cm * (1 in/2.54 cm) = Y in.
(Read this as "the number
of inches, times 2.54 centimeters per inch, equals the number of
centimeters"). Plug in the measurements you made.
(a) 21 in * (2.54 cm / 1 in) = 53.3 cm
(b) 15 in (2.54 cm / 1 in) = 38.1 cm
(c) 19 in * (2.54 cm / 1 in) = 48.3 cm
Now you
can see that
53.3cm
is greater than (>) 45cm wide
38.1cm
is less than (<) 39cm high
48.3cm
is greater than (>) 43cm deep
(2) Solution for microwave size: centimeters to inches (need microwave size
smaller than space)
X
cm * (0.3937 in / 1 cm) = Y in
(Read this as “the
number of centimeters, times 0.3937 inches per centimeter, equals the number of
inches.)
(a) 45cm * (0.3937 in / 1 cm) = 17.7 in
(b) 39cm * (0.3937 in / 1 cm) = 15.4 in
(c) 43cm * (0.3937 in / 1 cm) = 16.9 in
Now you
can see that
17.7in
is less than (<) 21in wide
15.4in
is greater than (>) 15in high
16.9in
is less than 19in deep
The microwave oven is too high for the space.
1.1 Conversions: Cancelling Units
The
units of measure must be accounted for in any conversion. The units are obvious
in simple conversions:
X
Km * (1 mi / 1.609 Km) = Y mi
Notice
that the km units cancel out when they occur in both the numerator and
the denominator, leaving mi as the final unit. Think of units in
fractions as numbers, in a sense. Where 3/3=1, simplifying the threes to 1,
km/km simplify and cancel out. But why bother? Well, cancelling out units also
allows you to make certain that you have the conversion set up correctly. For
example:
X Km
* (1.609 Km / 1 mi)
(an
incorrect conversion) expands to
X Km2 / mi
leaving
Km times Km per mi, or Km2/mi, obviously NOT what you
wanted! If the units are wrong, so will be the numerical calculation.
1.2
Conversions: Scientific Notation (see http://www.ieer.org/clssroom/scinote.html
for more)
What is
scientific notation? Scientific notation is a way of expressing very large or
very small numbers in a form that's short and easy to use. Consider the number
5,681,805,550,528.27. This was the U.S. National Debt on Jan 7, 2001 4:49:39
PST (from the following URL: http://www.brillig.com/debt_clock/).
If we round this number off to the second digit it still takes a lot of space:
5,700,000,000,000.00 dollars. However, if we remember that each place to the right
or left means multiplying or dividing by ten, then we can shorten the way the
number is written. 1000 is 10 times 10 times 10, or 103. Using this
transformation, 5,700,000,000,000.00 dollars becomes 5.7 x 1012dollars.
Starting from the left, count the places from the right of the first
number to the decimal point 5,700,000,000,000.00 (highlighted), then
move the decimal point to the position immediately to the right of the first
number, and use the number of places counted to give you the 'power of ten'.
Likewise,
the size of a Hydrogen atom may be written 0.000000012 meters. If we wanted to
determine how many Hydrogen atoms were present in 1m3 (cubic meter)
at the center of our sun, then the size of the Hydrogen atom would need to be
expressed in meters. Similarly, starting from the left, count the number of
places between the decimal point and the first number (including the number) 0.000000012
(highlighted), move the decimal point to the position immediately to the right
of the first number, and use the number of places counted to give us the 'power
of ten' (in this case a negative power because the number is less than 1). So,
0.000000012m becomes 1.2 x 10-8m.
We
simply reverse the process to convert to general format. For the first example,
a positive exponent, we move the decimal point 12 places to the right. For the
second example, a negative exponent, we move the decimal point 8 places to the
left.
2. SI (Systeme Internationale) Units and
Conversion Factors
SI Prefixes: Those commonly used (abbreviations in
parentheses)
|
|
Greater Than 1 |
|
|
Less Than 1 |
|
|
hecto- |
100 |
103 |
centi- |
0.01 |
10--2 |
|
kilo- |
1,000 |
103 |
milli- |
0.001 |
10-3 |
|
mega- |
1,000,000 |
106 |
micro- |
0.000001 |
10-6 |
|
giga- |
1,000,000,000 |
109 |
nano- |
0.000000001 |
10-9 |
Conversions
|
SI SYSTEM TO ENGLISH |
ENGLISH SYSTEM TO SI |
Length/Area: Common conversions, base unit = meter
(m)
|
1 meter (m) = 1.0936 yards =
39.37 inches (in or ") |
1 yard = [3 feet or 36 inches] = 0.914 meters |
|
1 centimeter (cm) =
0.3937 inches (in or ") |
1 inch = 2.54 centimeters |
|
1 kilometer (km) =
0.62137 miles (mi) |
1 mile = 1.609 kilometers |
|
1 hectare = 2.471 acres =
107,600 ft2 |
1 acre = [43,560 ft2] = 0.405 hectares |
Volume: Common conversions, base unit = liter (l)
|
1 liter (l) = 0.264 gallons (gal) =
[1.057 quarts (qt)] |
1 gallon = [4 quarts] = 3.785 liters |
|
1 milliliter (ml) = 0.03381 fluid ounces
(oz) |
1 fluid ounce = 29.57 milliliters |
Mass: Common conversions, base unit = gram (gm)
|
1 gram (gm) = 0.03527 ounces (oz) |
1 ounce = 28.35 grams |
|
1 kilogram (kg) = 2.205 pounds (lb) |
1 pound = 0.454 kilograms |
Other Common conversions
|
Energy: 1 joule (J) = 0.239 calories, (cal) [0.000948 British Thermal Units, btu] |
1 calorie = [0.0397 btu] 4.19 joules |
|
Pressure: 1 pascal
(Pa) = [0.01 millibars] = |
1 atmosphere = [14.7 pounds per square inch (psi)] 101300 Pascals [1013 millibars] |
|
Temperature: 0 Kelvin (K) = -273.15°Celsius,
(°C) = -459.7° Fahrenheit, (°F) |
0°C = [°C =
5/9(°F-32)] 32°F
= [°F = (9/5°C)+32] 273.1K [K =
°C +273.15] [°C =
K + 273.15] |
|
Astronomical Unit (AU) 1 AU=149,597,870km = 92,955,807 miles 1 AU is the average distance between the Earth and the Sun |
|
For more SI notation, units and conversions: http://www.chemie.fu-berlin.de/chemistry/general/si_en.html
For more conversions, including unusual and obsolete
ones, look here: http://www.convert-me.com/en/
The
notation is based on powers of base number 10. The general format looks
something like this:
N X 10x
where N= number greater than 1 but less than 10 and x=exponent of 10.
Placing numbers in exponential notation has several advantages:
- For very large numbers and extrememly small ones, these numbers can be placed in scientific notation in order to express them in a more concise form.
- In addition, numbers placed in this notation can be used in a computation with far greater ease. This last advantage was more practical before the advent of calculators and their abundance.
- In scientific fields,
scientific notation is still used. Let's first discuss how we will express
a number greater than 10 in such notational form.
3.1 Numbers Greater Than 10
- We first want to locate the decimal and move it either right or left so that there are only one non-zero digit to its left.
- The resulting placement of the decimal will produce the N part of the standard scientific notational expression.
- Count the number of places that you had to move the decimal to satisfy step 1 above.
- If it is to the left as it
will be for numbers greater than 10, that number of positions will equal x
in the general expression.
As an
example, how do we place the number
23419
in standard scientific notation?
- Position the decimal so that
there is only one non-zero digit to its left. In this case we end up with
2.3419
- Count the number of positions we had to move the decimal to the left and that will be x.
- Multiply the results of step 1 and 2 above for the standard form: So we have:
2.3419 X 104
3.2
How about numbers less than one?
We
generally follow the same steps except in order to position the decimal with
only one non-zero decimal to its left, we will have to move it to the RIGHT.
The number of positions that we had to move it to the right will be equal to
-x. In other words we will end up with a negative exponent.
Negative
exponents can be rewritten as values with positive exponents by taking the
inversion of the number.
For
example: 10-5 can be rewritten as 1/ 105.
Here is
an example to consider:
Express
the following number in scientific notation:
0.000436
- First, we will have to move
the decimal to the right in order to satisfy the condition of having one
non-zero digit to the left of the decimal. That will give us:
4.36
- Then we count the number of positions that we had to move it which was 4. That will equal -X or x = -4. And the expression will be:
4.36 X 10-4
3.3
What about numbers that are between 1 and 10?
In
those numbers we do not need to move the decimal so the exponent will be zero.
For example:
7.92
can be rewritten in notational form as:
7.92 X 100
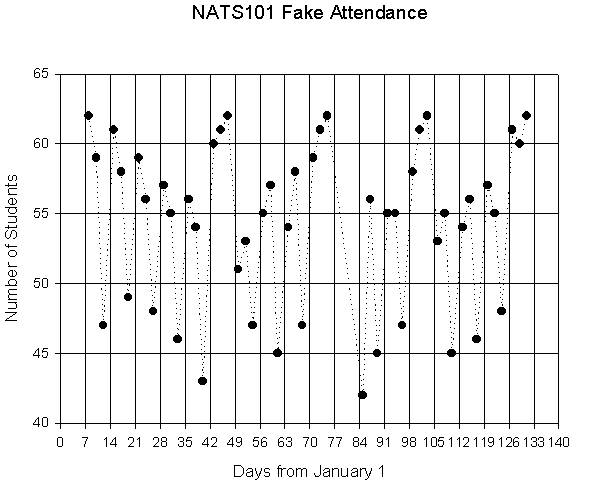
Graphs
allow us to present a lot of information in a small space, and to make it more
easily understandable. If we wanted to know how many days in a semester every
student showed up for class and what day had the lowest student attendance, we
might keep a running count for each class session. We could get the information
we want by looking down the table of numbers, but we can instantly see the
highs and lows if we make a plot of days against student attendance.
On the
next page I have produced a fake graph of student attendance for a class of 62
students, and added other information in the text that might be used to
interpret the graph. The scale across the bottom of the graph is time. The
scale along the left side of the graph is the number of students attending. Now
we find the place on the grid (lines) in between for each pair of numbers. For
instance, on day 29 there were 57 students in class. So the place where day 29
(moving up from the bottom scale) and 57 students (moving over from the left
scale)intersect becomes a unique dot on the graph telling us both values.
The
time axis (the x-axis), says "Days from 1/1/2001". The tick marks on
this axis are at 7 day intervals allowing any weekly information to be easily
seen. The axis for the number of students (the y-axis), says "Number of
Students" (duh). The tick marks for this axis mark off the student
attendance in fives.
From
the fake class syllabus I know that the dates for the exams were Feb 16th,
March 14th, April 13th, and May 11th. These
are Julian Days 47,73,103, and 131, respectively. Also, Spring Break was from
March 17th- March 25th. The lines between the datapoints
are not real, of course. No students were attending class when there was no
class. But these lines help us to see patterns in the data through time.
So what
interpretations can we make? We can immediately see a pattern related to the
time crossbars, placed at 7 day intervals. These students tended to come to
class on Mondays and Wednesdays, and miss Fridays. Also, notice the gradual dropoff
in attendance over the first five weeks of class, then a resurgence before the
first exam. In fact, this pattern recurs before each exam. Also, note that the
lowest attendance was on the Monday immediately after Spring Break. If we trace
up from the x-axis on this day (Day 85), and over from the y-axis, we see that
only 42 students showed up for class that day. The same procedure also tells us
that all students showed up on 5 days, those days being the first day of class
and each exam.
5. Periodic Table of the Elements
Atoms
are composed of neutrons (neutral charge), protons (positive charge) and
electrons (negative charge). The neutrons and protons are located in the
nucleus and the electrons revolve around the nucleus. When the number of electrons
and protons in an atom are equal, the atom is “neutral”; if they
are not equal, the atom is “ionized”. The Periodic Table of the Elements ranks the
chemical elements in order of increasing number of protons, known as the atomic
number.
6.1.
Conversion to and from Scientific Notation (a page on scientific notation
is available at the end of this document) Also, you can check you answers at http://www.quickmath.com/.
Use the
information provided on the pages on conversions at the end of this homework to
convert these numbers (fill in the blanks).
|
General Notation |
Scientific Notation |
|
13,500,000 Pascals (Pa) |
|
|
|
4.5 x 10-9 liters (l) |
|
0.000000071 kilometers
(km) |
|
|
|
1.23 x 1018 kilograms (kg) |
2.
Graph Interpretation
Below
are two graphs. The first graph presents the carbon dioxide (CO2)
concentration as measured at
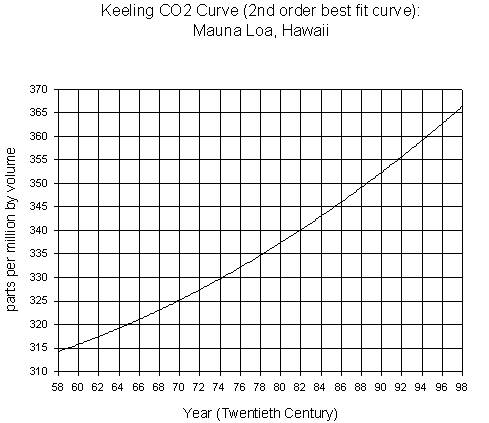
The second graph presents CO2 concentration reconstructed, at
irregular time intervals, from gas trapped in an ice core collected at Taylor
Dome,
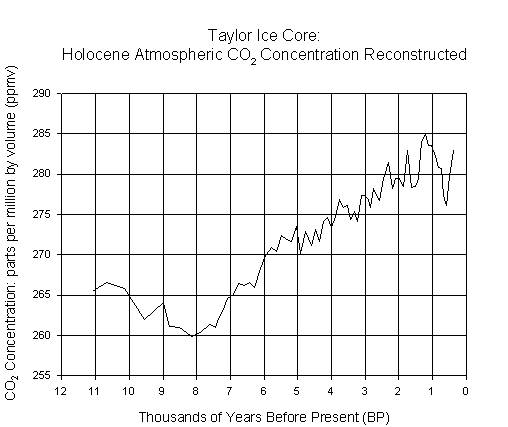
6.2.
Answer the following questions about these graphs.
(2a) Years
are the unit plotted on the x-axis on these two graphs. The most recent data
points on the first graph are on the right side. Is this true for the second
graph?
(2b)
The y-axes on both graphs present CO2 concentration in parts per
million by volume. What is the range of concentrations presented on the first
graph (to the closest 5ppmv)? What is the range on the second graph (to the
closest 5ppmv)?
(2c) In
what year do we see the highest CO2 concentration in the first
graph? What is the concentration (to the nearest 5 ppmv)?
(2d) At
what time do we see the lowest CO2 concentration in the second graph
(to the nearest thousand years)? What is the lowest concentration (in ppmv)?
What is the difference (mathematically) between the highest concentration in
the first graph and the lowest concentration in the second graph? (show your
calculations)
(2e)
What is the change in CO2 concentration between 1958 to 1967
(10 years), early in the history of the measurements, and 1989 to 1998 (10
years), the latest years on the first graph? (show your calculations)
6.3.
Periodic Table of the Elements. What chemical elements correspond
to the following atomic numbers (put down the 2-letter chemical symbol and the
full name):
1 =
_______________________________ 12 = ______________________________
16 =
_______________________________ 14 = ______________________________
11 =
_______________________________ 7 = ______________________________
6 =
_______________________________ 79 = ______________________________
Last
modified 8/29/07.
Back to NATS101 home page.